AI in healthcare workflows 2026 is less about buying a shiny model and more about removing friction in the steps that quietly drain staff time: documentation, messages, authorizations, routing, and handoffs.
If you work inside a U.S. hospital, you probably feel the same tension every budget cycle, leadership wants “AI wins,” frontline teams want fewer clicks, compliance wants less risk, and IT wants fewer one-off tools that don’t integrate.
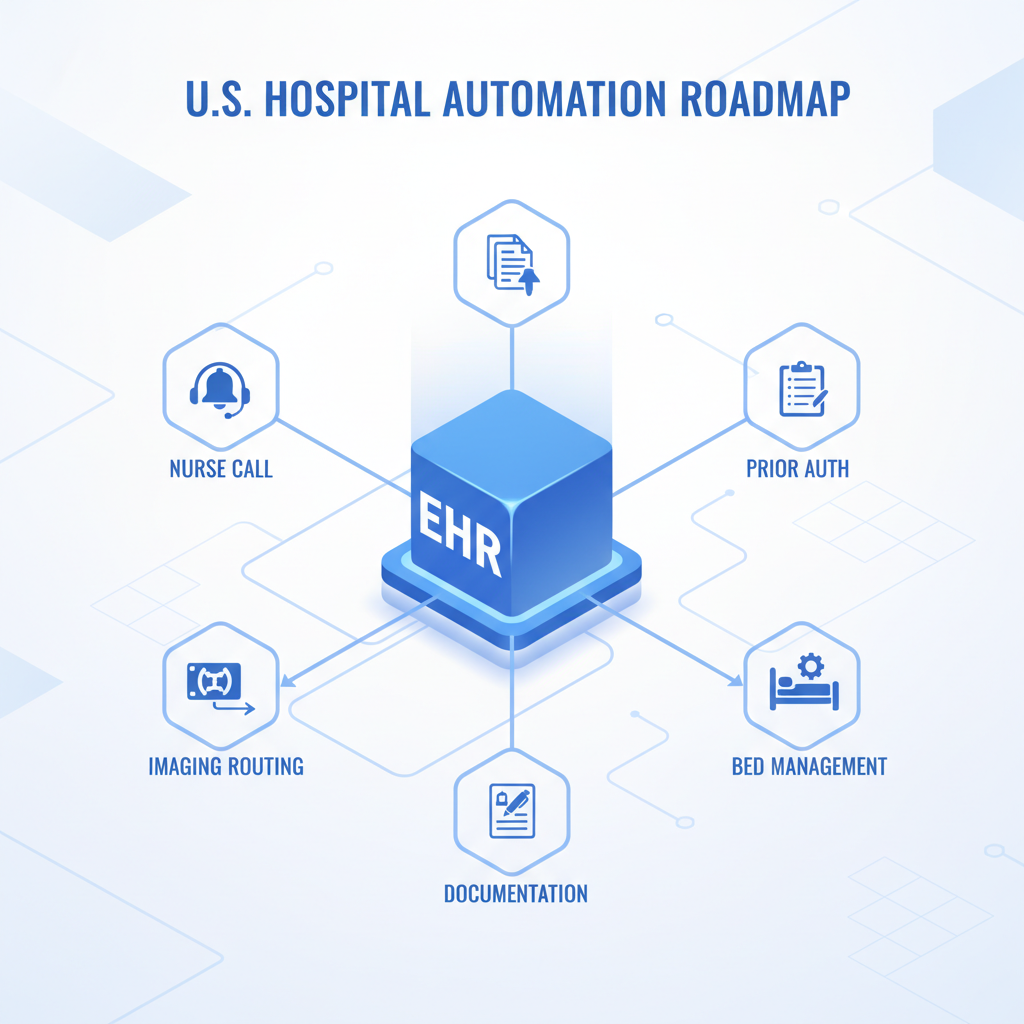
This guide focuses on what to automate first, what to delay, and what to avoid, with practical checkpoints you can use in governance meetings. It also calls out where “automation” must still keep a human in the loop, because patient safety and regulatory reality matter more than demo-day performance.
What “automation” should mean in a hospital (and what it shouldn’t)
In many hospitals, the fastest AI value comes from workflow orchestration, meaning the system routes work to the right person, at the right time, with the right context, and documents what happened. Not replacing clinicians, not guessing diagnoses.
In 2026 conversations, the most useful framing is: automate administrative and coordination load first, then assist clinical decisions where evidence, auditability, and guardrails are strong.
- Good automation targets: repetitive tasks, standardized criteria, predictable routing, high-volume documentation, denial-prone steps with clear rules.
- Risky automation targets: autonomous triage without supervision, black-box recommendations without traceability, tools that can’t write back to the EHR cleanly.
According to the U.S. Food & Drug Administration (FDA), clinical software using AI can fall under medical device oversight depending on intended use, so “just a workflow tool” can shift categories if you let it influence clinical decisions without the right controls.
What to automate first: a practical priority stack for 2026
If you need a “what do we do Monday” sequence, prioritize based on volume × friction × measurability. The themes below map to your keyword areas, but the real win is choosing 2–3 that share infrastructure and governance.
1) Ambient clinical documentation tools (start here if clinician burnout is the headline)
Ambient documentation can be a high-visibility win, but only if you treat it as a workflow project, not a microphone project. The goal is faster note completion with fewer after-hours edits, while keeping clinician authorship clear.
- Deploy in 1–2 specialties where note structure is relatively consistent.
- Define what counts as “final” and who validates problem lists, orders, and diagnoses.
- Require edit tracing and source attribution so compliance can audit how text entered the chart.
2) Prior authorization automation (start here if denials are your pain)
Prior auth is a workflow maze: documentation gathering, payer rules, status chasing, peer-to-peer scheduling. Automating the “chase” and “packet assembly” parts often delivers immediate operational relief.
- Standardize clinical criteria templates per service line before automating.
- Automate status checks, missing-doc alerts, and submission packaging.
- Keep a human checkpoint for medical necessity narratives and edge cases.
According to the Centers for Medicare & Medicaid Services (CMS), payers and providers operate within specific coverage and documentation expectations, so your automation should improve completeness and timeliness, not invent justification.
3) Revenue cycle AI for claims accuracy (quiet ROI, fewer “avoidable” denials)
Many hospitals chase big clinical AI while leaving money on the table in coding edits, claim scrubs, and mismatch detection. This is often safer than clinical decision automation because the outputs are more auditable.
- Use AI to flag coding-documentation mismatches and missing modifiers.
- Automate work queues by likelihood of denial and aging risk.
- Measure impact in denial reasons and rework time, not just “clean claim rate.”
Clinical workflow automation that pays off fast (without putting you in a safety bind)
Clinical workflow automation tends to deliver the best returns where it reduces interruptions, clarifies responsibility, and improves handoffs. The trick is staying on the right side of safety and scope.
AI triage and nurse call optimization (optimize routing, not autonomous clinical judgment)
“Triage AI” sounds like diagnosis, but the safer win is message prioritization and routing: who should respond, how fast, and with what context.
- Normalize call reasons and message categories so the model has stable inputs.
- Route to role-based pools (charge nurse, unit clerk, rapid response) with escalation logic.
- Track false-urgent vs missed-urgent events as core safety metrics.

Predictive patient flow and bed management (use forecasts to trigger actions)
Forecasting discharges and admissions is not new; what’s new in AI in healthcare workflows 2026 is integrating forecasts into operational triggers: EVS dispatch, transport scheduling, bed assignment pre-work, and discharge barrier checklists.
- Start with a single campus and compare forecasts to actuals by unit type.
- Convert predictions into tasks with owners, not just dashboards.
- Keep override capability obvious, because surge events break patterns.
Imaging workflow prioritization and routing (reduce bottlenecks without overpromising)
Radiology often benefits from AI that prioritizes worklists and routes studies to the right subspecialty or location. This can improve turnaround time without claiming to “diagnose.”
- Use AI to flag potential critical findings for earlier review, with clear labeling.
- Automate study routing based on modality, history, and staffing.
- Define downtime workflows so radiologists aren’t stranded when AI is unavailable.
Interoperability and FHIR-enabled orchestration: the part everyone underestimates
Hospitals usually don’t fail at pilots, they fail at scaling, and the blocker is almost always integration. If your AI tools can’t reliably read context and write back results, staff end up doing “human middleware.”
FHIR-enabled orchestration means using standards-based APIs to move tasks, context, and status between systems, EHR, imaging, call systems, RPM platforms, and RCM tools. It’s not glamorous, but it prevents the tool sprawl that makes operations brittle.
- Insist on write-back patterns that preserve source, timestamps, and responsible user.
- Define a canonical patient and encounter context to reduce mismatches.
- Govern vendor access, logging, and retention like any other clinical system integration.
According to HL7, FHIR is designed to enable consistent health data exchange, which is why orchestration built on standards typically scales better than custom point-to-point interfaces.
Medication safety and pharmacy operations AI: high impact, higher governance
Medication workflows are safety-critical, so the bar for automation should be higher. Still, there are valuable targets that support pharmacists instead of replacing judgment.
- Order verification support: highlight unusual doses, duplicate therapy risk, renal dosing mismatches, with explanations clinicians can audit.
- Inventory and dispensing operations: predict shortages, optimize restocking routes, reduce expired stock risk.
- MAR and administration workflow: flag timing conflicts or missing documentation for follow-up.
In many cases, teams get better outcomes by focusing on exception management rather than trying to automate every order review step.
Remote patient monitoring workflow integration: value depends on who “owns the inbox”
RPM programs stall when alerts pile up and nobody is clearly responsible for follow-up. The technology works, but the workflow collapses under alert fatigue.
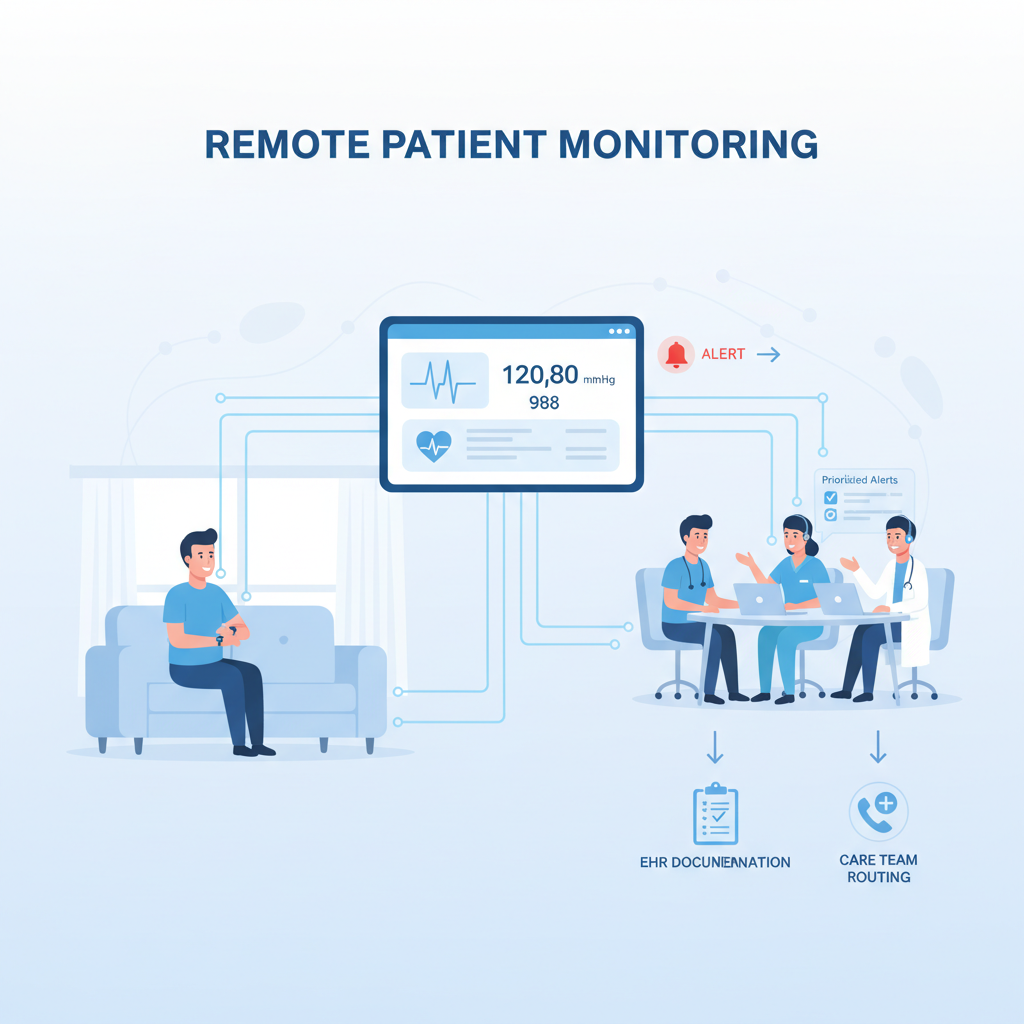
For AI in healthcare workflows 2026, the practical move is to integrate RPM into existing care pathways, not bolt it on as a parallel program.
- Define alert thresholds and escalation rules per condition and risk tier.
- Route alerts into role-based queues, not individual hero clinicians.
- Close the loop by writing outcomes back into the EHR as structured data when possible.
Quick self-assessment: are you ready to automate this workflow?
This is the checklist that prevents you from scaling chaos. If you answer “no” to several items, start with standardization and instrumentation, then re-evaluate.
- Clear inputs: do we have consistent categories, templates, and definitions?
- Clear owner: who is accountable when the automation is wrong or ambiguous?
- Measurable outcome: can we track time saved, turnaround time, or rework reduction?
- Safe fallback: what happens during downtime or low-confidence outputs?
- Integration path: can the tool read context and write results where staff already work?
- Auditability: can we trace who approved, edited, and finalized key outputs?
What to avoid (or delay) even if vendors pitch it hard
Some projects look impressive and still disappoint, usually because they create risk or require behavior change that hospitals can’t sustain.
- Autonomous clinical triage without tight supervision: high liability, hard to validate across populations, and easy to misuse operationally.
- “One model for everything” platforms: often end up as expensive shelfware when integration and governance aren’t specific.
- Replacing structured documentation with free text: it can make downstream coding, quality reporting, and care coordination harder.
- Automation without change management: if staff training and workflow redesign are missing, the tool becomes a new inbox.
According to the Office of the National Coordinator for Health Information Technology (ONC), interoperability and trustworthy data exchange depend on consistent standards and governance, so avoid solutions that require “special exceptions” to connect.
Implementation playbook: 90-day steps that usually work
Hospitals that get real traction tend to run small, measurable deployments with strong governance, then scale once the workflow proves stable.
- Pick one workflow and one metric: for example, note completion time, denial rework hours, or imaging turnaround time.
- Map the handoffs: identify where tasks bounce between roles, that’s where automation helps.
- Define guardrails: confidence thresholds, escalation rules, and “no automation” scenarios.
- Instrument from day one: logs, audit trails, and exception reasons should be non-negotiable.
- Run parallel for a short window: compare AI-assisted vs standard workflow before full cutover.
A simple decision table: match use cases to risk and readiness
Use this table in steering committees to keep conversations grounded in operations, not hype.
| Use case | Typical ROI speed | Risk level | What to prove before scaling |
|---|---|---|---|
| Ambient clinical documentation | Medium | Medium | Note quality, edit burden, compliance audit trail |
| Prior authorization automation | Fast | Low–Medium | Submission completeness, cycle time, payer-specific exceptions |
| Revenue cycle AI for claims accuracy | Fast | Low | Denial reason reduction, coder rework time, explainability |
| Nurse call routing optimization | Medium | Medium | Escalation accuracy, missed urgent events, staff adoption |
| Predictive bed management | Medium | Medium | Forecast accuracy by unit, operational trigger success rate |
| Imaging prioritization and routing | Medium | Medium–High | Turnaround time, QA review findings, downtime workflow |
| Medication safety and pharmacy ops AI | Medium | High | Alert precision, override patterns, adverse event monitoring plan |
| RPM workflow integration | Slow–Medium | Medium | Alert burden, follow-up closure rate, documentation write-back |
Key takeaways for hospital leaders
- Automate coordination first: routing, packaging, status checks, and documentation assembly often beat “clinical magic.”
- Integration is the moat: interoperability and FHIR-enabled orchestration determine whether pilots scale.
- Safety needs design: guardrails, audit trails, and fallbacks are part of the product, not paperwork.
- Pick fewer bets: two workflows that share plumbing can outperform six disconnected tools.
Conclusion: where to start this quarter
For most U.S. hospitals, the most defensible starting points in AI in healthcare workflows 2026 are prior authorization automation, revenue cycle accuracy, and ambient documentation in a controlled rollout, because the outcomes are measurable and the governance path is clearer.
If you want one action to take next, choose a single workflow, set a 90-day metric, and insist on integration and auditability before expanding, the “cool” demo becomes a durable operational capability.
FAQ
What should U.S. hospitals automate first with AI in 2026?
Usually the fastest wins come from documentation support, prior auth packaging and status chasing, and claims accuracy workflows, because they reduce rework without pushing AI into unsupervised clinical decisions.
Is clinical workflow automation the same as clinical decision-making?
No, and mixing them causes trouble. Workflow automation routes tasks and assembles context; clinical decision-making influences diagnosis or treatment and typically needs stricter validation and oversight.
How do we evaluate ambient clinical documentation tools safely?
Look for edit traceability, clear clinician sign-off, strong PHI handling, and a realistic plan for exceptions, like noisy environments or complex visits where the draft note may be misleading.
Will AI triage replace nurses or reduce staffing needs?
In many cases it works better as prioritization and routing support. Staffing impacts vary widely, and hospitals often see value as fewer interruptions and faster responses rather than straightforward headcount reduction.
What does FHIR-enabled orchestration actually change operationally?
It reduces “copy-paste integration,” so tasks, statuses, and documentation can flow between systems with less custom glue code. That makes scale and governance more realistic, especially across multiple sites.
How can we prevent RPM alerts from overwhelming care teams?
Set thresholds by risk tier, route alerts to pooled queues, and measure closure rates. If alerts don’t reliably convert into documented actions, the program becomes noise fast.
When should we involve compliance, legal, or patient safety teams?
Early, ideally before go-live criteria are finalized. If a tool could influence clinical decisions, touches PHI in new ways, or changes documentation authorship, it’s worth getting formal review and documenting guardrails.
If you’re trying to pick 2–3 automations that won’t collapse under integration debt, it can help to start with a workflow map, then shortlist vendors and build a simple FHIR-first architecture plan before the pilot begins.
